|
The history of the ABCD nationwide
and worldwide audit programme
The establishment of the ABCD nationwide audit programme
on N3 - the latest version of NHSnet, represents the culmination of a
national audit programme which has been evolving and becoming
increasingly larger and more sophisticated since 2004. Below the
national audits are listed in reverse chronological order:
2022 - ABCD worldwide Omnipod audit
Following the success of the first audit of a device, the FreeStyle
Libre audit, this audit is the fourth device related audit and has
launched in March 2022. All users of the Omnipod system are invited to
join the audit. Historic as well as current data may be entered and we
are collecting data on all Omnipod systems including before and after
switches to the latest Ominipod Dash system.
Click here for more information and to join the audit.
2021 - ABCD DTN-UK nationwide closed loop audit
This audit was launched in July 2021 to coincide with the
NHS England pilot of broader criteria for closed loop use in England
– namely the use of closed loop systems in people already on pump
therapy with FreeStyle Libre and a HbA1c≥69mmol/mol. The audit is not
restricted to those in the
NHS England pilot and all users of closed loop systems are invited
to encouraged to join.
Click here for more information and to join the audit.
2021 – Nationwide Oral Semaglutide audit
The nationwide Rybelsus® audit launched in June 2021. All users of oral
semaglutide are invited to join the audit.
Click here for more information and to join the audit.
2020 - ABCD COVID-19 & Diabetes: UK national audit
Launched during 2020 in response to the COVID-19 pandemic.
The methodology has been published in the British Journal of Diabetes.
Click here for more information. During 2021 collaborations were
established with groups in France, Spain and New York.
2020 - Worldwide audit of
testosterone deficiency in men with type 2 diabetes
Launched in December 2020. All who treat such patients with
testosterone are invited to join.
Click here for more information and to join the audit.
2020 - International Do-It-Yourself Artificial Pancreas System
(DIY APS) audit.
All who have patients who use DIY-APS are invited to join.
Click here for more
information and to join the audit.
2019 - Nationwide Semaglutide audit
The nationwide Ozempic® audit launched in February 2019. All users of
Semaglutide are invited to join the audit.
Click here for more information and
to join the audit.
2018 - Nationwide FreeStyle Libre audit
The nationwide FreeStyle Libre audit launched in August 2018 and
represents our first audit of a device. All users of FreeStyle Libre are invited to join the audit.
Click here for more information and
to join the audit.
2017 - Nationwide IDegLira audit
The nationwide IDegLira® audit launched in April 2017. All users of
IDegLira, liraglutide and degludec are invited to join the audit.
Click here for more information and
to join the audit.
2017 - Nationwide empagliflozin audit
The nationwide Invokana® audit launched in March 2017. All users of empagliflozin are invited to join the audit.
Click here for more information and
to join the audit.
2016 - Nationwide canagliflozin audit
The nationwide Invokana® audit launched in January 2016. All users of
canagliflozin are invited to join the audit.
Click here for more information and
to join the audit.
2014 - Nationwide insulin degludec audit
The nationwide Tresiba® audit launched in November 2014. All users of
insulin degludec are invited to join the audit.
Click here for more information and
to join the audit.
2014 - Nationwide dapagliflozin audit
The nationwide Forxiga® audit launched in October 2014. All users of
dapagliflozin are invited to join the audit.
Click here for more information and
to join the audit.
2014 - Nationwide exenatide QW audit
The nationwide Bydureon® audit launched in April 2014. All users of
exenatide QW are invited to join the audit.
Click here for more information and
to join the audit.
2014 - National audit of DKA management
This audit aims to assess diabetic ketoacidosis management (DKA)
management across the UK following the introduction of the latest
version of the JBDS DKA guideline (see
guidelines 2a and 2b ). For more information
click here.
2012-2014 - IDOP and ABCD care homes
audit
The Institute of Diabetes for Older People (IDOP) in association with
ABCD has led a national stakeholder audit of diabetes care in care
homes. Responses from more than 2000 care homes in England revealed
important gaps in screening for diabetes, hypoglycaemia care, and the
training and education of care staff in the area of diabetes. The
results of this audit have been passed to the Care Quality Commission to
assist them in developing inspection processes for care homes.
The report, launched in
June, 2014, is available on both the IDOP and ABCD websites.
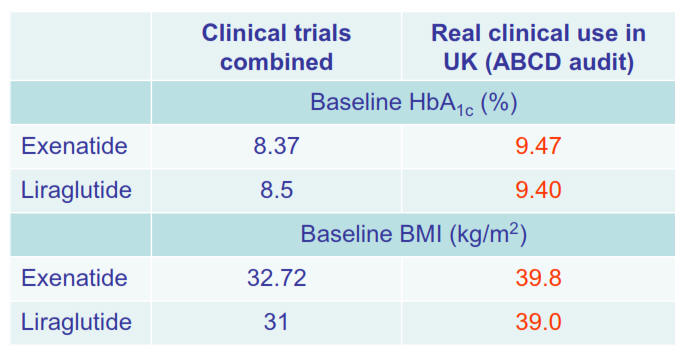
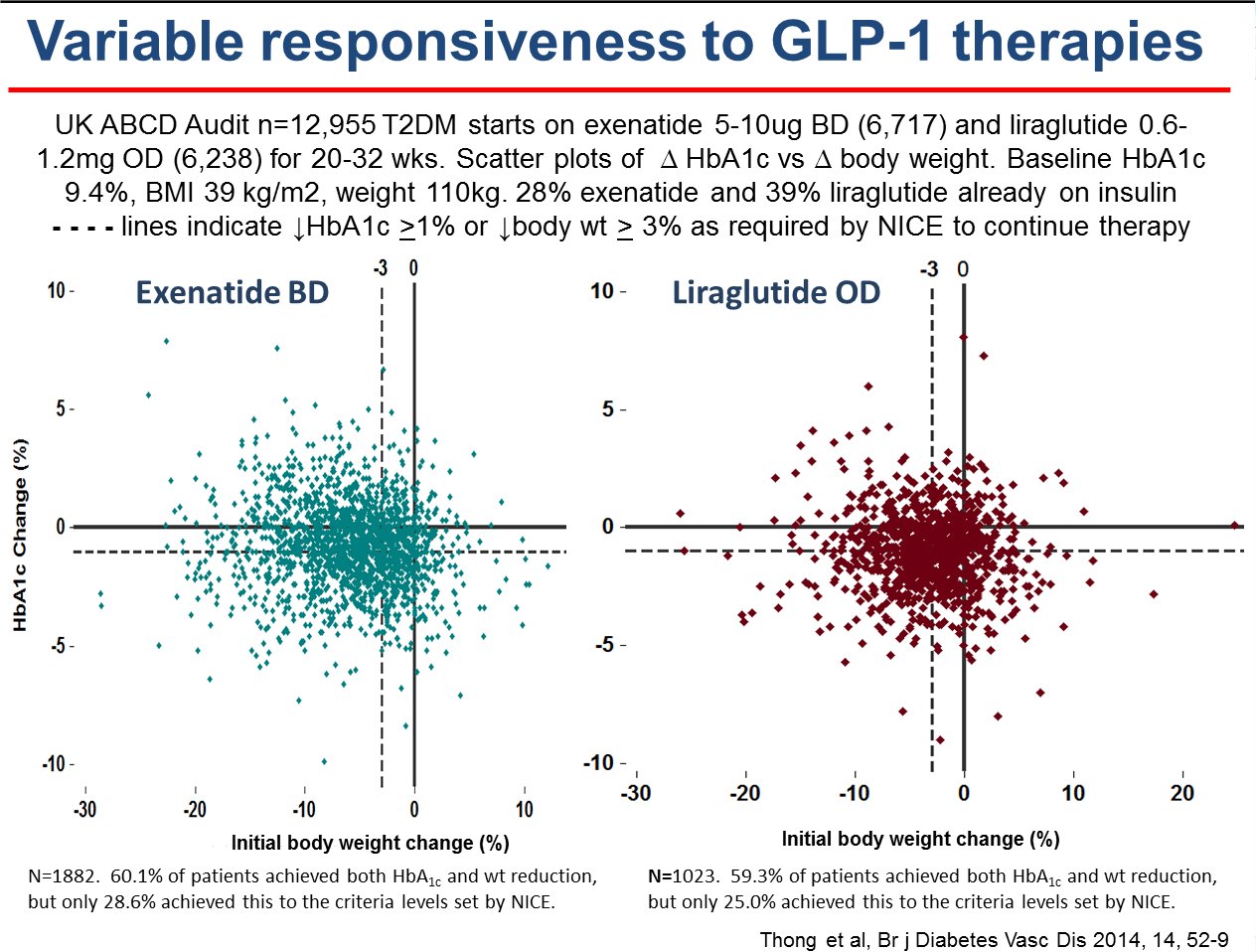
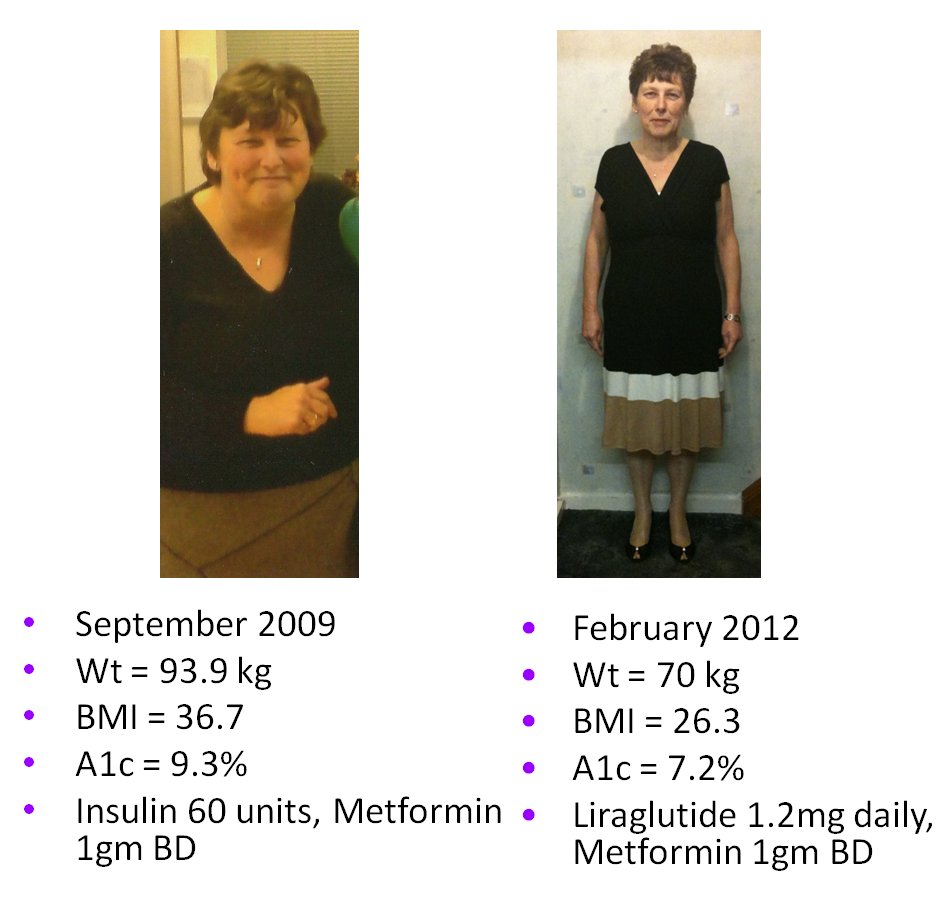
2009-2014 - Nationwide liraglutide audit
- ongoing since 2009
The
ABCD nationwide liraglutide audit is ongoing and now has over 6000
patients. There have been
several presentations at national and international meetings and
there are papers
published and being written. The audit tool was upgraded in 2012 to
become secure online/web-based. All users of liraglutide are invited to
join the audit.
2009-2014 - ABCD Scottish audit of
inpatient diabetes care - ongoing since 2009
An audit of Inpatient Diabetes Care across NHS Lothian: The
effectiveness of the use of information technology, the Scottish Patient
Safety Programme and ‘Think Glucose’.
2011-2013 - ABCD/JDRF/Diabetes UK Insulin
Pump Audit
The joint ABCD/JDRF/Diabetes UK Insulin Pump Audit for the UK surveyed
diabetes centres in the UK, to establish basic data on numbers and type
of insulin pump starts and follow up, and availability and use of CGMS.
The audit was
published in May, 2013, and revealed that the proportion of people
in the UK who live with type 1 diabetes and use a pump. Publication was
accompanied by a
press release from JDRF and Diabetes UK, giving their reaction to
the audit’s findings and headlined “UK lagging behind Europe for use of
insulin pumps”.
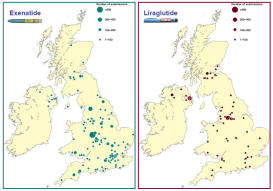
2008-2011 - Nationwide exenatide audit
The ABCD nationwide exenatide audit represented a landmark in that
it attracted so many contributors and hence data on a large number of
patients. 6717 patients
(2007-2009), 315 contributors, 126 centres. Several papers are now
published
with more still being written and there have been
many presentations at
national and international meetings.
2007 - Nationwide exubera audit
This audit of the inhalable insulin
exubera was launched in 2007 but was never completed because
exubera was taken off the market.
2005-2006 Nationwide audit of glitazone
use with insulin
A nationwide audits undertaken in 2005 and
presented as a poster at the ABCD meeting in Glasgow in 2006. The
audit found that in extreme insulin resistance some, but not all,
patients responded to the combination.
2005-2006 - Nationwide audit of triple
OHA use in insulin avoidance
A nationwide audits undertaken in 2005 and
presented as a poster at the ABCD meeting in Glasgow in 2006. The
audit demonstrated that the addition of glitazones to metformin and
sulphonylureas was often very effective and allowed insulin to be
avoided or postponed.
2005 - Nationwide audit of gestational
diabetes screening
For this audit questionnaires were sent to all ABCD members asking them
to describe how in their locality patients were screened for GDM, the
diagnostic criteria and subsequent management and clinical targets.
The findings were
published in QJM in 2008 and showed that standards for GDM screening
and management vary significantly across the UK..
2004 - Nationwide audit of glargine in
pregnancy
This audit charted the outcomes on 109 babies from 115 women treated
with glargine insulin during pregnancy. The findings were
published in Diabetic Medicine in 2008 and suggested that the use of
glargine in Type 1 diabetes during pregnancy was not associated with any
unexpected adverse maternal or fetal outcome. |