|
|
The ABCD nationwide liraglutide audit
About the ABCD nationwide
liraglutide audit
This audit was established in the wake of the success of the original
ABCD nationwide exenatide audit. Learning from the exenatide audit a
considerably improved tool for the audit was created and the audit was
launched in the Autumn of 2009. The audit had a number of
objectives.
Though data collection has ceased during 2018, analysis of the data
continues.
Over 10 years of audit
2019 marked the 10th anniversary of the launch of the audit and saw the
publication of the 9th paper.
In 2020 a 10th paper is in press. As well as
10 published papers, there have also been
17 published abstracts,
eight oral presentations and
17 poster presentations.
What we learned from the exenatide
and liraglutide audits
After the audit had been active for 5 years, at the ABCD autumn meeting
in 2014, Dr Ryder summarised the main findings from the ABCD nationwide
exenatide and liraglutide audits and
the slides from that presentation
are available for viewing. In summary, the findings over the 10 years
since the audit was launched have been:
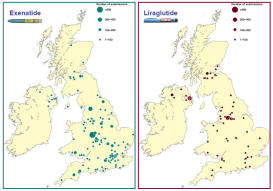
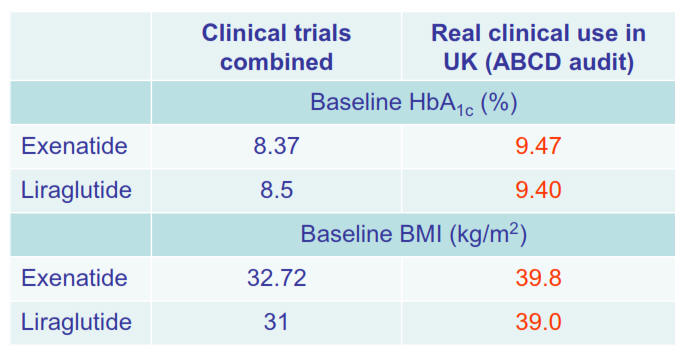
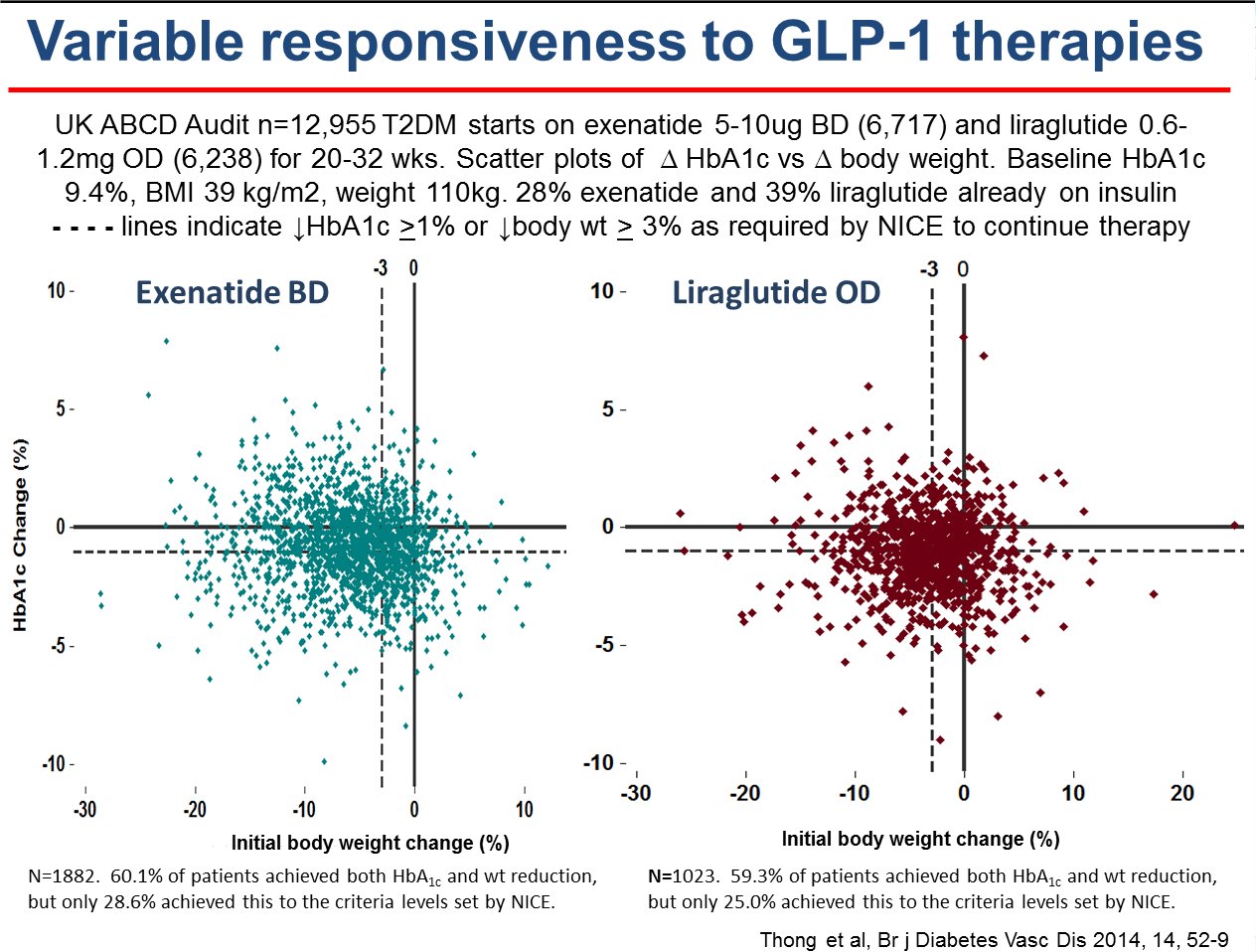
• The patients treated with exenatide or liraglutide in real clinical practice are much
heavier and with much poorer glycaemic control than in clinical trials
of these agents; nevertheless, the agents have proven to be very
effective.1
• Exenatide and liraglutide were used outside NICE guidelines in
substantial numbers of patients and were found to be effective in
outside NICE guidelines. In particular, the agents were used with
insulin (40% in the nationwide liraglutide audit) with good effect in
many patient.2
• Off licence use of exenatide with insulin was found to be
safe and effective in real clinical practice, a reduction in insulin
dose frequently occurred, weight fell and 1 in 6 patients were able to
come off insulin.3,4
• An important safety issue was uncovered in that some clinicians
attempted to stop insulin when starting exenatide in order to stay
within license. This led to harm to the patient in some instances - for
example there are 11 reported cases of ketosis or diabetic ketoacidosis
- 7 of these occurred to patients who stopped insulin at the time of
exenatide initiation. Analysis of audit data allowed us to recommend
that when starting exenatide in an insulin-treated patient, not to stop
the insulin but rather to tail the insulin off during treatment if
response to treatment allowed.5,6
• When concerns were raised in 2013 in a TV documentary and in the
British Medical Journal that incretin therapies might cause pancreatic
damage, we were able to publish data suggesting that in the ABCD audits
there is no evidence of such a side effect and that most cases of acute
pancreatitis in the ABCD exenatide and liraglutide audits had other
causes for acute pancreatitis, in particular gall bladder disease.7,8,9,10,11
• Many patients with a professional driver’s licence who would lose
their jobs if they went onto insulin, were able to avoid insulin, and
maintain similar glycaemic outcomes and keep their jobs by using
exenatide or liraglutide.12,13
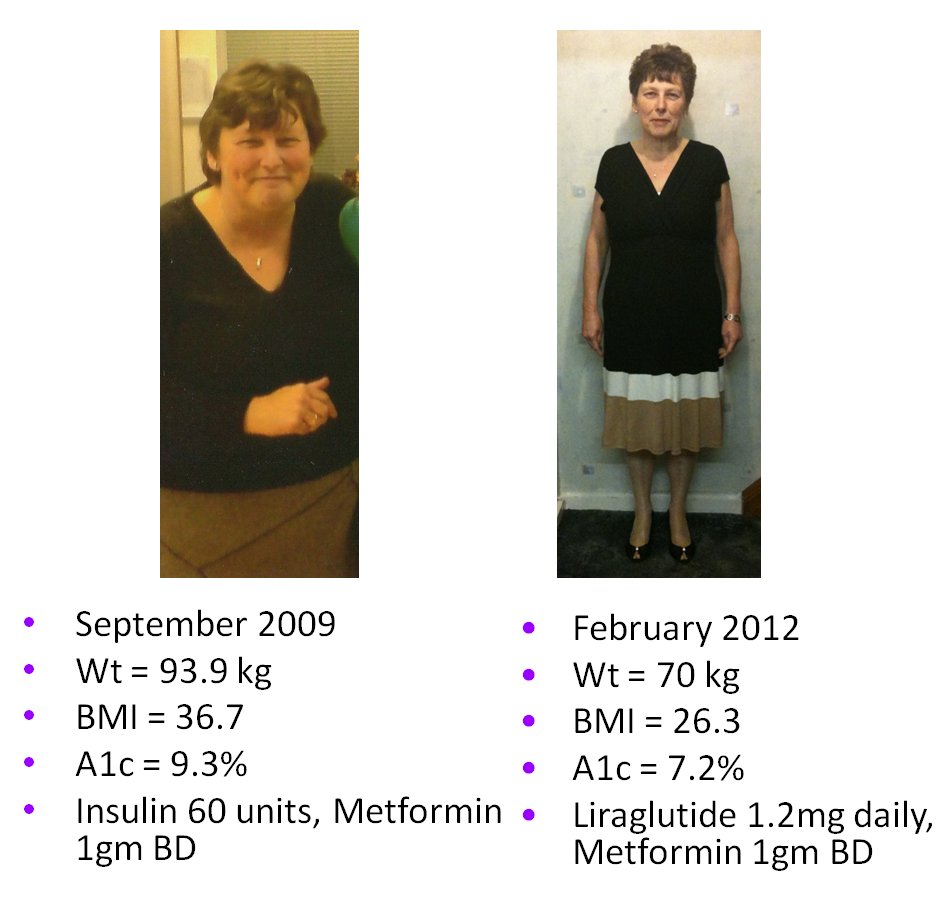
• Liraglutide was found to be safe and effective among patients with
moderate renal impairment, even though that was an exclusion for use at
the time.14,15
• Liraglutide use was found to be associated with a reduction in alanine
aminotransferase levels (an index of fat in the liver) when they are
elevated.16
• Though initially liraglutide was not licensed for use with insulin and
in due course it was licensed only for use with insulin determir, it was
found in the audit to be effective with all the common insulin regimes,
whether basal, basal-bolus or biphasic.17
• Exenatide and liraglutide may be less effective at improving glycaemic
control amongst non-insulin treated South Asians.18
• Long duration of diabetes and insulin use both predict reduced
response to liraglutide, with insulin use being the strongest predictor.19
• Improvements in HbA1c and weight were seen when switching from
exenatide and DPP4 inhibitors to liraglutide.20
• Older age and non-metformin use were associated with more significant
gastro-intestinal side effects leading to discontinuation of liraglutide
treatment.21,22
• In occasional patients the nausea, vomiting or diarrhoea was so severe
that they developed transient acute kidney injury. No other new safety
issues were uncovered.23,24
• Starting liraglutide was found to reduce 10-year cardiovascular risk
as assessed by the UKPDS risk-engine.25,26
Further
information
Further enquiries may be made to the ABCD nationwide audits
database administrator of the project,
Melissa Cull |
Liraglutide audit objectives - 2009
First visit data entry form used in the audit
Follow up visit data entry form used in the audit
Nationwide exenatide audit
Papers, abstracts, presentations, posters from the audit
Further
information- contact us
Main ABCD homepage
|